SECTION 6 - REGULATED WASTE
REGULATED / MEDICAL WASTE
COMPANY USED FOR REMOVAL AND DISPOSAL:
________________________________________________________________
Address: ________________________________________________________
________________________________________________________
Phone #: ________________________________________________________
Method used for disposal: ___________________________________________
WHERE RECORDS ARE KEPT:
________________________________________________________________
PERSON IN CHARGE OF RECORD KEEPING:
________________________________________________________________
It is the responsibility of this person above to read and understand the requirements for Regulated Medical Waste according to OSHA and local regulations. The information in this section is supplementary to the information your State provides.
If person in charge of record keeping changes or medical waste company changes, write in new information below and date:
_______________________________________ ______________
_______________________________________ ______________
_______________________________________ ______________
EMPLOYEES (TITLE) WHO HANDLE WASTE AS PART OF THEIR JOB DESCRIPTION:
______________________________________________
______________________________________________
______________________________________________
______________________________________________
______________________________________________
POLICY: The above employees will be trained on safety when handling medical waste. This training will be part of initial job orientation and annual (combined with OSHA Bloodborne safety training).
WHAT IS REGULATED MEDICAL WASTE?
“A regulated medical waste is any solid, liquid or semi-liquid waste, generated in the diagnosis, treatment, (e.g., provision of medical services), or immunization of human beings or animals, in research pertaining there to, or in the production or testing of biologicals. In addition, regulated medical waste is further defined at N.J.A.C. 7:26-3A.6 and includes, among other things: culture and stocks of infectious agents, human pathological waste, human blood and blood products, used and unused sharps including syringes, contaminated animal waste, and specialized isolation waste. Regulated medical waste that is both treated and destroyed is no longer regulated medical waste. (Regulated medical waste is treated when the biological character is changed so as to substantially reduce or eliminate its potential for causing disease, and it is destroyed when it is processed so that it is no longer generally recognizable as medical waste.) Regulated medical waste that is treated but not destroyed, or destroyed but not treated, is still considered regulated medical waste.”“Regulated Medical Waste” is classified as follows: Classification
1. Cultures and stocks,
2. Human pathological waste,
3. Human blood/blood products,
4. Sharps - hypodermic needles, syringes, pasteur pipettes, blood vials, scalpel blades, immunization vials, culture dishes, broken glass (only if contaminated),
5. Contaminated animal waste,
6. Special isolation waste - bio-level4 CDC, and
7. Unused sharps. Items that are caked with dried blood or potentially infectious materials AND are capable of releasing these materials when handled will be considered regulated medical waste.
Medical waste tracking forms, and required logging and reporting forms can be obtained from your medical waste disposal company or the State DEP, EPA or DOH - Division of Solid Waste Management, that oversees medical waste disposal. The address and phone number the State Department that is in charge of Regulated Medical waste is:
______________________________________
______________________________________
______________________________________
Phone # _______________________________
CLASSES OF REGULATED MEDICAL WASTE
Class 1: CULTURES AND STOCKS OF INFECTIOUS AGENTSCultures and stocks of infectious agents and associated biological including:
cultures from medical and pathological laboratories; cultures and stocks of infectious agents from research and industrial laboratories; wastes from the production of biological: discarded live and attenuated vaccines; and culture dishes and devices used to transfer, inoculate and mix cultures.
Class 2: HUMAN PATHOLOGICAL WASTES
Human pathological wastes, including tissues, organs, and, body parts, and fluids that are removed during surgery or autopsy or -other medical procedures, and specimens of body fluids and their containers
Class 3: HUMAN BLOOD AND BLOOD PRODUCTS
Liquid waste, human blood, products of blood, items saturated and / or dripping with blood that are now caked with dried human blood; including Serum, plasma, and other blood components and their containers which were use-d or intended for use in patient care testing and laboratory analysis., or the development of pharmaceuticals. Intravenous bags are also-included in this category.
Class 4: USED SHARPS
Sharps that have been used in animal or human .patient care or treatment in medical research or industrial laboratories including- hypodermic needles, syringes (with or without the attached needle), pasteur pipettes, scalpel blades, blood vials, needles with attached tubing and culture dishes ( regardless of presence of infectious agents), Also included are other types of broken or unbroken glassware that were in contact with infectious agents..
Class 5: ANIMAL WASTE
Contaminated animal carcasses, body parts and bedding of animals that were known to have been exposed to infectious agents during research (including research in veterinary hospitals), production of biologicals or testing of pharmaceuticals
Class 6: ISOLATION WASTE
Biological waste and discarded materials contaminated with blood, excretion, exudates or secretions from humans who are isolated to protect others from certain highly communicable diseases or isolated animals known to be infected with highly communicable diseases
Class 7: UNUSED SHARPS
Unused, discarded hypodermic needles, suture needles, syringes, and scalpel blades
MEDICAL WASTE DISPOSAL POLICY
Disposal of all infectious waste shall be in accordance with Federal, State and local regulations (outside of N.J. N.Y, Pennsylvania and Florida, you are responsible for getting information from your state and/local DEP). All infectious waste shall be placed in closable, leak-proof containers or bags that are color-coded, labeled or tagged. If is likely that outside contamination of the container or bag will occur, a second leak-proof container or bag must be placed over the outside of the first container.
ALL medical waste containers will be labeled with the generators name, address and license number/ID number (if applicable).
Disposable syringes, needles, scalpel blades and other sharp items shall be placed in puncture resistant containers for disposal. Container must be properly labeled. Containers will not be filled more than 213 full.
Puncture-resistant sharps containers shall be easily accessible to workers and located in areas where they are commonly used.
Lab specimens of body fluids shall be transported in a container that will prevent leaking and disposed of in accordance with regulatory requirements.
List regulated wastes specific to this facility and area located below:
___________________________________________________________
___________________________________________________________
___________________________________________________________
Location where regulated waste is stored until picked up:
___________________________________________________________
List any additional policies here (eg. transport, segregation):
___________________________________________________________
___________________________________________________________
___________________________________________________________
___________________________________________________________
WASTE DISPOSAL - FACILITY POLICY
Facility waste is segregated at the point of origin into categories that include but are not limited to:1. Recycled waste (e.g., paper, aluminum, glass) Employees are encouraged to recycle in appropriate containers, which are located in the following area: _____________________________.This facility re cycles the following items (1) _________________________ (2)______________________ (3)______________________Recycled items are collected by ___________________________________ on a (daily, weekly, monthly) basis.
2. Regular Trash Regular trash is waste that poses no health or environmental risk. It is disposed of through routine waste facilities/companies in appropriate containers. The following company or town/city picks up this waste: ___________________________________________________
3. Infectious / biohazardous “red bag” waste All infectious/biohazard waste shall be placed in closable, leak-proof containers or bags that are color-coded, labeled or tagged. All red bags will be labeled with the generators name, address and ID number (if applicable). Infectiousbiohazard waste in the state of________________________________ and the city of________________________ must be handled appropriately, according to Federal, State, and local laws. Infectious waste must be picked up by a licensed medical waste company and then either incinerated, autoclaved, ground or decontami nated before disposal in a sanitary landfill. Bulk blood, suction fluids, excretions, and secretions may be carefully poured down a drain if connected to a sanitary sewer where permitted. Sanitary sewers may also be used to dispose of other infectious waste capable of being ground and flushed into the sewer, where permitted. Biohazard containers are kept upright throughout use and are routinely checked by __________________________________________________ biohazard red bag waste is then placed in and then removed/stored when two thirds full. All leak-proof biohazard containers and stored in ____________________________It is then disposed of by (company) ______________________________________________(frequency) _______________
4. Needles/sharps
Disposable syringes, needles, scalpel blades and other sharp items shall be placed in puncture resistant “sharps” containers for disposal. Containers will not be filled more than 2/3 full. All containers will be labeled with the generators name, address and ID number (if applicable). Puncture resistant sharps containers shall be easilyaccessible to workers and located in areas where they are commonly used. All sharps and glassware are disposed of immediately (or as soon as possible) in containers that are closable, puncture-resistant, leak-proof on the sides and bottom, and appropriately labeled with a biohazard label. Location of sharps containers:
___________________________ ___________________________
___________________________ ___________________________
___________________________ ___________________________
5. Chemical Waste
Chemical waste (e.g. bulk liquid chemical, chemotherapy) will be disposed of according to local and federal regulations. Write policy here:
___________________________________________________________
___________________________________________________________
___________________________________________________________
___________________________________________________________
6. Record keeping
The following records of waste disposal will be kept for a minimum of 3 years:
State license/ID number (if applicable), medical waste transporters license number, generator pick up (tracking) form and waste disposal/destruction confirmation form.
PROCEDURE FOR PREPARING MEDICAL WASTE
Medical waste generated by this facility must be properly packaged, labeled, and marked before being transported to an off-site facility for treatment or disposal.PROCEDURES
1. Medical waste generated by this facility will be placed in a container or combination of containers that are rigid and leak resistant, impervious to moisture, strong enough to prevent tearing or bursting under normal handling, and securely sealed.2. Sharps (e.g., needles, razor blades, broken glass, syringes, lancets, scalpel blades, etc.) will be packaged in a rigid container resistant to punctures.
3. Fluids will be poured into a container resistant to breaking and equipped with a tightly fitting lid or stopper.
4. A water-resistant label will be affixed to all containers or packages of untreated medical wastes.
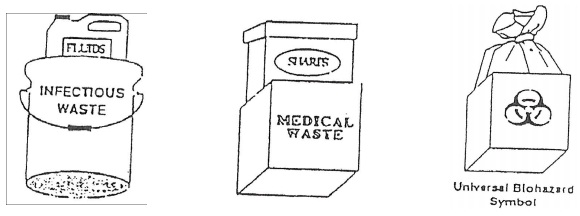
5. When there is inner and outer packaging, or containers, each container (inside and outside) will be labeled “Infectious Waste” or “Medical Waste” or display the universal Biohazard Symbol as illustrated below:
6. Red plastic bags, when used as inner packaging for untreated waste, need no labels. However, the outer packaging or container will be identified by the proper label or symbol.
7. Before shipment, all packaging-inner and outer-will be marked with indelible ink or with a water resistant identification tag. Each inner container, if used, will show the name and address of this facility (or identifying numbers).
* A training guide for packaging medical waste follows this page
WHAT IS OVER-CLASSIFIED MEDICAL WASTE?
OSHA lists what is classified as regulated waste (see page 6-3). If the facility generates a waste that is potentially infectious but does not fall into a regulated category the facility must decide how to handle this waste. Infectious or contaminated waste that does not fall into the regulated category is called over classified waste”. There are two disposal options for this waste. It is the responsibility of the person in charge of medical waste to check with local, county and state regulations before making the decision on how to dispose of questionable materials. The follow serves as a guideline:Option 1: If put in a red bag, the bag must be picked up by your medical waste carrier. If your State charges the facility by the pound for a medical waste licence, you can keep the weight of this waste separate since it is not technically regulated waste. This weight can be deducted from the total weight picked up by the medical waste disposal company so it would not be counted toward the medical waste licencing fee.
Option 2: If the town and county has no regulations concerning disposal of nonregulated medical waste, you can put this waste in a regular (not red) trash bag. This trash must be segregated in the facility and MAY NOT be mixed with “clean” trash. If this option is used - the trash can (not bag) this waste is stored in must have a lid and the can or lid must be labeled with a bio-hazard or a infectious waste label. The reason the can must be marked is to protect the employees and warn other people that they should not put their hand into this waste receptacle and wear gloves when tying up bag for removal. There are two disposal options:
(1) the bag can be put out with the regular trash
(2) the bag can be put in the biohazard waste storage box, keeping track of the weight if it effects the cost of the medical waste licencing fee.
It is your responsibility to make sure this option is acceptable before putting it out
Option 2: to your regular trash for pick up.
If you are un-sure whether an item is “regulated” or “over-classified” call your State department of medical waste and ASK!
CONSIDERATIONS FOR DRAIN DISPOSAL OF CHEMICALS
1. Certain chemicals are permissible for drain disposal.2. Local sanitary sewer district will be contacted to determine what is and what is not acceptable.
3. Facility’s waste water flow rate should be calculated to document any chemical concentration at time of entry into sanitary sewer system.
4. Drain system should eventually flow to waste water treatment plant.
5. Only those chemicals reasonably soluble in water are suitable for drain disposal. A compound is considered water soluble if it dissolves to the extent of at least 3%. These compounds will be flushed with at least 100 volumes of excess water.
Some exceptions should be noted:
A. Organics with boiling points less than 50 C.
B. Hydrocarbons, halogenated hydrocarbons, nitro compounds, mercaptans, and most oxygenated compounds that contain more than five carbon atoms (E.g.: Freon).
C. Organics that are explosives such as azides and peroxides.
Check with your State DEP, water company or medical hazardous waste disposal company if you are unsure of regulations for drain disposal for certain chemical or biohazardous liquids.
HELPFUL HINTS
Designate one person to keep records. Records will include: contract and licence numbers of medical waste pick up company, pick up receipt (usually white form is given at time of pick-up) and disposal (usually gold or yellow) confirmation form (should be returned to facility within 2-3 weeks of pick up). If you have state licencing, a copy of this licence should be put in with medical waste records. Check that it is renewed annually. Sharps containers will be placed in a “safe” area. If wall mounted the top should be slightly below eye level (52-56 inches). Pediatric offices must wall mount. Storage medical waste boxes with red bag liners will be in a “non-patient” area.
Get a copy of your state regulations. Read the regulations and call the DEP or government agency that oversees this, if you have any questions.
Obtain a copy of your medical waste companies licence numbers. Make sure their licence is current. Obtain new information each year. This is your responsibility!
Keep all records organized in a divided file or notebook. Sections should include State regulations, medical waste disposal company information, tracking form, logs and correspondence.
Check with your town or private pick-up company before putting “over-classified” medical waste out with the regular trash.
Check with your local water department before pouring any body fluids or chemicals down the drain.
REGULATED MEDICAL WASTE PROCEDURES AND LAWS SPECIFIC TO YOUR STATE EACH FACILITY MUST ADD THESE LAWS
 Hours:
Hours: