HAZARD DEFINITION
A toxic or hazardous substance regulated under the OSHA standard(s) is any substance which has the capacity to produce personal injury or illness to man through ingestion, inhalation, or absorption through anybody surface.
BLOODBORNE PATHOGEN STANDARD
This standard was proposed in May 1989 and became effective in March 1992. The standard was developed in hope of reducing the amount of occupational related cases of Hepatitis B, HIV and other potentially infectious diseases among health care workers. The standard covers over 20 infectious diseases. The facility will follow a written exposure control plan. The plan will be reviewed and upgraded annually. The law provides guidance by addressing the following areas: Developing an exposure control plan that identifies all tasks and procedures where exposure may take place.
Job classifications - determining and documenting which employees are at risk (form for this is found in the record keeping section of this manual).
Implementing - Universal and Standard Precautions, engineering controls, work practice controls and use of personal protective equipment.
HBV vaccine program
Post-exposure evaluation and follow-up policies
Infectious waste disposal policies
Use of biohazard labels and bags
Housekeeping, decontamination and laundry practices
Employee safety education and training
Record keeping documents
The risk of exposure to potentially infectious body fluids should never be underestimated. Common fluids included are blood, blood products, semen, vaginal secretions, cerebrospinal fluid, synovial fluid, pleural fluid, peritoneal fluid, pericardial fluid, amniotic fluid, saliva (in dental setting) and any other potentially infectious materials.
ELEMENTS OF THE EXPOSURE CONTROL PLAN
The following items are all part of the exposure control plan. The following information is a review of the requirements and where to find or document these requirements. Employer will identify tasks and procedures where occupational exposure may occur.
Identify employee exposure determination by filling out the “Job Exposure Classification” form (page 8-4) found in the “Record Keeping” section of this manual.
Develop policies for job specific procedures and tasks that have an exposure risk. Policies will be documented by the safety officer using the “Safety for Procedures Performed” form found in this chapter (pg. -18) of the manual.
Offer the HBV vaccine (within 10 working days of hire) and follow up blood test to confirm immunity status to employees in job classification 1 &.2. Employees will sign the “Hepatitis B Vaccination” form found in the “Record Keeping” section (page 8-7) of this manual.
Take and document a comprehensive Safety Program (prior to job task assignment with potential exposure then annually) that covers the topic listed in the “Safety Training” section of this manual.
Put in place the appropriate work practice controls, engineering controls and use of personal protective equipment. General policies are listed on the “Work Practice and Engineering Controls” pages found in this chapter. Facility should add task specific policies. NOTE: If facility has not complied with items applicable on these pages, the date of compliance should be noted next to the item on list.
Biohazard labels and signs will be used to communicate hazards to employees. Guidelines and policies for biohazard labeling is found in the “Labels” section of this manual.
Personal Protective equipment will be supplied, replaced and maintained by the employer in sizes that insure proper fit. General guidelines for what type and when to wear PPE is found in this chapter. Job task specific PPE is listed in the individual “Safety for Procedures Performed” policies.
Housekeeping practices, facility cleaning and laundry practices will be documented and followed according to the schedule found in this chapter of the manual.
Medical waste will be disposed of as outlined in the chapter on medical waste and in accordance with OSHA and State law
All exposure incidents will be recorded as an injury. A “Post-Exposure” incident report form is found in the “Record Keeping” section of this manual. Instructions on how to follow up a body fluid exposure are in this chapter (page 4-24 to 4-26). Employee medical records are confidential. Injury records are to be kept for 30 years after the person leaves employment.
The exposure control plan will be reviewed and upgraded as needed or annually. This can be documented using the form found on page 1-3.
FACILITY EXPOSURE CONTROL PLAN CHECKLIST
UNIVERSAL PRECAUTIONS / STANDARD PRECAUTIONSPOLICY: Employees will follow Universal and Standard Precautions at all times. Both the Centers for Disease Control and The American Hospital Association recommend that ALL patient’s blood and body fluids be handled as if infectious. Universal precautions, as defined by the CDC, are a set of precautions designed to help prevent transmission of Hepatitis, the AIDS virus (HIV) and other bloodborne pathogens. Body fluids listed under universal precautions are human blood and blood products, semen and vaginal secretions, pericardial, peritoneal, pleural, amniotic, synovial and cerebrospinal fluids, unfixed tissues and organs from humans (living or dead), saliva in dental procedure and any body fluid or clinical specimen containing visible blood. Basic aseptic techniques must be used to limit blood contact can prevent HBV, HCV and HIV transmission in the health care setting. In September 1997 the CDC published new infection control guidelines and introduced “Standard Precautions’. Standard precautions incorporate the major features of universal precautions but added precautions for ALL body fluids, secretions and excretions (except sweat), regardless of whether or not they contain visible blood (as stated in universal precautions). It also includes precautions for non-intact skin, mucous membranes, guidelines for airborne diseases (<5um), droplet precautions (> 5um) and contact precautions (e.g by hand contact)
Basic universal and standard precautions include:
1. Use barrier protection routinely to prevent skin and mucous membrane contamination with blood or other body fluids.2. Hands will always be washed before and after contact with patients. Hands will be washed even when gloves have been used. If hands come in contact with blood, body fluids or human tissue, they will be immediately washed with soap and water.
3. GLOVES will be worn when contact with blood, body fluid, tissues and contaminated surfaces is anticipated.
4. Gowns, lab coats or plastic aprons are indicated if blood splattering is likely.
5. MASKS and PROTECTIVE GOGGLES will be worn if aerosolization or splattering are likely to occur (e.g. certain dental and surgical procedures, wound irrigations, postmortem examinations and bronchoscopy).
6. To minimize the exposure when performing emergency mouth-to-mouth resuscitation, mouth pieces, resuscitation bags, or other ventilation devices will be strategically located and available for use in areas where resuscitation may occur.
7. Sharp objects will be handled in such a manner to prevent accidental cuts or punctures. Used needles will not be bent, broken, reinserted into their original sheath or unnecessarily handled. They will be discarded intact immediately after use into an “sharps” needle disposal box which will be readily accessible (placed in all clinical areas, including patient rooms). All needle stick accidents, mucosal splashes or contamination of open wounds with blood or body fluids will be reported immediately.
8. Body fluid spills will be cleaned up promptly with a hospital grade disinfectant solution or a 1 : 10 dilution of bleach.
9. All patients’ blood specimens will be considered biohazardous. All specimens must be placed in plastic zip-lock bags for transport.
10. Linen soiled with blood or body fluids from ANY patient will be bagged and handled cautiously.
11. Non-disposable articles contaminated with blood or body fluids will be bagged and labeled using biohazard bags for decontamination and reprocessing.
12. Handle laboratory instruments, especially needles and scalpel blades with extreme caution.
13. Use biological safety hoods for procedures that have a high potential for generating droplets.
14. Fill evacuation tubes, vial, and bottles by using their internal vacuum only.
15. Use mechanical pipettes.
16. Decontaminate work area and equipment if contaminated with blood, other body fluids or potential droplet contamination.
17. Patient-Care equipment will be discarded or properly disinfected and/or sterilized.
* Additional policies are found throughout this chapter 3
SPECIAL PRECAUTIONS FOR PHLEBOTOMY
All patient specimens will be handled as potentially infectious. No specimen will be considered a higher risk than another.
Employees will always wear gloves - there are no exceptions.
Additional protection such as laboratory coat, eye protection, face shields or masks will be available for use if splashing, spraying or splattering is expected.
When possible - blood will be collected using vacuum tube(s) rather than syringe to avoid unnecessary transferral of specimen.
The employer and staff will determined by evaluation if the use of safety needles and/or safety holders will reduce number of needle stick injuries among the staff. Based on this evaluation safety needles will be purchased and used. Training will be provided on how to use.
Needles will not be bent, broken, recapped or removed from holder unless the employer demonstrates that no alternative is feasible.
If vacuum tubes are filled from a syringe, use the internal vacuum only.
Tourniquets will be disposable or disinfected after use if visible soiled.
All specimens must be transported to the lab or stored in the lab will be placed in a secondary container (eg. phlebotomy tray, test tube rack, plastic bag) to minimize accidental contamination, spillage or breakage. All secondary containers will be labeled with a biohazard label and disinfected regularly.
On June 12, 2002 OSHA released a position on the removal of contaminated needles from blood tube holders. The release stated (quote), “Removing con taminated needles and reusing blood tube holders can expose workers to multiple hazards. We (OSHA) want to make it very clear that this practice is prohibited in order to protect workers from being exposed to contaminated needles.”
WORK PRACTICE CONTROLS
Work practice controls reduce the likelihood of exposure to potentially infectious materials by ensuring that procedures performed in the facility are performed using the safest technique possible. The fundamental work practice control is Universal Precautions. The health care worker must recognize the potential risks and make the decision of what necessary steps will be taken to maximize protection. risks can be minimized or eliminated by following simple work practice rules.POLICY: The following work practice controls will be followed:
Avoid hand-to-face contact while working
Do not eat, drink, apply cosmetics, handle contact lenses or smoke in patient care areas
Do not put food or drinks in lab refrigerators or freezers
Wash hands when gloves are removed
Do not answer phone or use computer while wearing gloves.
Do not recap needles (unless using “scooping techniques” or mechanical means)
Cover corks with gauze when opening vacutainers
Never reach into waste containers that might have contaminated waste
No mouth pipetting
Decontaminate work areas as needed
Ask coughing patients to wear a surgical mask or turn their heads and use a tissue
Let falling objects (e.g. needle, tube of blood) fall
SLOW DOWN - most accidents happen when you are rushing!
ADD ADDITIONAL WORK PRACTICE CONTROLS SPECIFIC TO YOUR PRACTICE HERE:
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
ENGINEERING CONTROLS
An engineering control is a type of technology or device that isolates, removes or reduces the potential exposure hazard of a bloodborne pathogen. The combination of engineering and work practice controls are the recommended method of minimizing exposure to potentially infectious body fluids or materials. These methods are preferred because their effectiveness does not depend on the individual’s decision of whether or not to use them properly. These controls are examined, evaluated,upgraded, maintained, or replaced on a regular schedule by the employer to ensure safety.POLICY: The following Engineering Controls have been put in place:
Hand washing facilities near or in all work areas. If sink is not available, antiseptic hand cleaner/towelettes will be available.
Sharps containers located in or near work areas where procedures are performed.
Leak proof storage containers for blood and body fluid specimens. (e.g. stoppered test tubes, zip-lock bags, test tube racks)
Red biohazard bags for disposal of all infectious (non-sharps) waste. The bag and receptacle will be marked with a biohazard sign.
Eye wash station (within 100 ft. or 10 sec. from area where exposure may occur)
Spill kit (for body fluid spills)
The following are additional engineering controls. Check the ones applicable to your facilities safety policies. Add additional ones you may have purchased.
—— Safety and/or self-sheathing needles (document evaluation - pg.4-13 to 4-16)
—— Centrifuge has lid or tubes have caps
—— Mechanical/automated pipettes will be used in all laboratory procedures.
—— Splash shield
—— Bio-safety hood
—— Secondary containers used for transport of needles or specimens
—— Plastic capillary tubes
Other ________________________________________
ENGINEERING CONTROLS
ENGINEERING/WORK PRACTICE CONTROLSSAFETY NEEDLE AND SATETY DEVICE EVALUATION
POLICY: As part of our facilities compliance with OSHA’s Bloodborne Pathogen Standard, the employer and/or safety officer and employees will evaluate and institute engineering controls that include safety needles and safety devices. The purpose of these controls will be to eliminating or minimizing employee exposure to potentially infections diseases. The needles and devices selected will be appropriate to procedures being performed. The selection of safety needles and devices will be based on a combination of the facilities history of exposure incidents and the potential for exposure (if no such history exists). The evaluation will be documented. Employee training on the use of controls selected will be provided and documented. The safety needle program will be reviewed and revised annually to include the addition of new technologies that have been considered or put in place in the facility. A11 sharps injuries will be documented in accordance with OSHA regulations.
Explanation: The Bloodborne Pathogen Standard has always mandated the use of engineering controls (section d-2-1) and work practice controls to reduce or eliminate employee exposure to bloodborne pathogens. Items such as sharps containers, sinks to wash hands, eye wash stations, mechanical pipettes, splash shields have always been accepted as valuable engineering controls. Since the Bloodborne regulations were passes in 1991, many new safety technologies have become available. Examples would be safety needles (e.g. self-sheathing needles & needleless IV connectors) and plastic capillary tubes. The 11/5/99 Bloodborne Pathogen directive states (section D-2) that these new technologies must be considered and requires the facility to evaluate exposure incidents and determine if a safety device could have eliminated or reduced the chance of the accident from happening. On November 6,2000 the Needlestick Safety Act (HR5178) was signed into law by President Clinton. OSHA published the final rule in the January 1 8, 2001 Federal Register. The new law became effective April 18, 2001. Simply stated - if a safety needle or safety device will reduce, remove or isolate the hazard, they must be used. The employer will invoive the employees with the evaluation and selection of appropriate safety devices. Devices evaluated must be FDA cleared. Note that just because a device is FDA cleared it does not guarantee the device will be effective. Employees must still follow standard precautions, use work practice controls and wear personal protective equipment. Medical supply companies should carry a variety of safety needles and devices to evaluate. When evaluating safety needles and devices look for the following:
1. Safety feature provides a barrier between the hands and the needle after use. The safety feature allows the workers hands to remain behind the needle at all times.
2. The safety feature is an integral part of the device and not an accessory.
3. The safety feature is in effect before disassembly and remains in effect after disposal to protect users and trash handlers.
4. The safety feature is as simple as possible, requiring little or no training to use.
If the facility is inspected by OSHA, the compliance officer (OSHA inspector) will determine through interviews or observation of work whether sufficient engineering controls and work practices are used. The compliance officer will review exposure incident reports. The compliance officer will determine if there were occasions where injuries occurred during the same procedure, using the same equipment, in the same location or among similar employees and determine whether engineering or work practices were implemented that could have prevented or minimized injuries. The compliance officer will investigate if the employer has instituted alternative en gineering controls in the areas where exposure injuries have occurred. The employer will be cited for the failure to investigate and implement engineering or work practice controls. If the facility is using a safety device, but the compliance officer feels another device would be more effective, the compliance officer will document this and consult with the regional bloodbome pathogen coordinator to determine if a violation exists. The facilities investigation of safety needles and devices must be documented. The employer must document that employees were trained on the proper operation and use of the safety needle and/or device. The compliance officer will review the facilities investigation and determine if training was provided or lack of training resulted in the failure to use such controls.
Policy, for performing a safety needle evaluation:
1. The employer and/or safety officer will select products to be evaluated based on procedures performed and analysis of exposure injuries. Use form #1.2. Evaluate three to four (or more) products. Products will be evaluated by individuals (employees) who will use the product. Document employees performing evaluation.
3. Instructions on use the safety device/needle and evaluations forms (form #2) will be provided.
4. Products will be tested in a simulated patient procedure (e.g. injection pads, oranges, training dummies).
5. Each evaluator will rate the system using question sheet provided.
6. Evaluator will express concerns that may not be covered on the evaluation form.
7 . The employer or safety officer will review the evaluations and document this review (form #3).
8. The product(s) that scored best will be purchased.
9. Training will be provided to employees that will use the products and were not involved in the evaluation process.
10. The safety officer will annually evaluate the safety needle and device program - purchasing new products if indicated.
DOCUMENTATION OF COMPLIANCE WITH THE SAFETY NEEDLE LAW
___________________ Date Safety needle/device program was implemented___________________ (Check if applicable) Safety needle/device program evaluated
but not implemented. Reason is document on evaluation form #3 or document separately
Program was re-evaluated on : By (name)
_________________________ _______________________________
________________________ _______________________________
________________________ _______________________________
________________________ _______________________________
________________________ _______________________________
SAFETY PROTOCOLS FOR PROCEDURES
(STANDARD OPERATING & EXPOSURE CONTROL PROCEDURES)
Each medical facility has special safety procedures that are unique to that particular practice. These procedures combine work practice controls, engineering controls, personal protective equipment, disinfecting procedures and medical waste. The fundamental procedure would be using universal precautions. Safety policies reduce the likelihood of exposure to hazards. Many times the risks can be eliminated by changing the way a procedure is performed. Make copies of the following SAFETY PROCEDURE FORM. Fill in the information for any procedure that involves exposure to potentially infectious body fluids. Do not limit these policies to only infection control - this form can be used for safety for other hazards such as chemicals and X-ray as well. Common examples are listed below.
Check the ones you do and add to this list.
Acupuncture IUD & PessariesAllergy Testing IV Set Up
Aspiration of Breast Cyst Lab Tests (performing)
Aspiration (needle - general) Laser
Amniocentesis Laser for hair removal
Artificial Insemination Leep
Biopsies Lesion/Abscess Incision
Bone Marrow Lumbar puncture
Cast removal Medical Waste (transporting)
Cleaning of suction machine Myringotomy
Chemical peels Nuclear Stress Test
Chemotherapy (administering) Paracentesis
Circumcision Removal of nasal packing
Colposcopy Shaving of body hair
Cryosurgery Sigmoidoscopies
Cystoscopy Spirometry
D&C Staple removal
Debridement Surgical Procedures (major)
Dressing change/wound care Surgical Procedures (minor)
Electromyograph Suturing
Endoscopy Suture removal
Endometrial Biopsy Terminations
Finger or Heel Stick Thoracentesis
Freezing with nitrogen Trigger point injections
Hysteroscopy Vaginal exams (PAP and KOH)
Injections (not live vaccine) Vasectomy
Inoculation (live vaccine) Venipuncture
Intramuscular Injections X-ray
Note: Policies above and additional policies are available from POC for $3.00 each. Call for list.
HAND WASHING
Most infectious agents found in the clinical facilities are spread by direct contact or respiratory exposure. Good hand washing and adherence to universal precautions will help break the chain of transmission and produce a safe work environment. The employer must provide adequate hand washing facilities. If a sink is not available in a patient care room, an anti-bacterial hand cleaner will be made available for immediate use until the employee can get to a nearby sink.POLICY: During daily activities, your hands come in contact with many organisms and that can transmit infections. Frequent hand washing is essential for both your protection and your patients. Hands will be washed:
If contaminated by blood, body fluids, or tissues.
After every patient contact.
After removal of gloves and other protective wear or equipment.
Before leaving the clinical work areas.
Before eating, drinking, smoking, applying makeup, or changing contact lenses outside of the lab, and after using lavatory facilities.
Before all other activities that entail hand contact with mucous membranes or a break In the skin.
Periodically during the day when routinely handling and testing body fluids.
The wearing of rings and bracelets will be avoided because they might interfere with a thorough hand washing. They also make it more difficult to don gloves!
PROPER HAND WASHING PROCEDURE
1. Wet hands and apply a small amount of an antiseptic soap (3-5 ml).2. Vigorously lather hands and rub together for at least 15 seconds.
3. Wash well between the fingers, under nails and up the wrists.
4. Rinse well with a moderate stream of water in a downward motion.
5. Dry with a paper towel and use the towel to turn off the faucet. Dry skin adequately to avoid dermatitis.
Alcohol hand rinse or disinfecting foam products may also be used for quick decontaminating the hands whenever clean running water is not available. However, these products should not be used as a substitution for hand washing.
Frequent hand washing can be very damaging to the skin’s normal protective mechanisms by damaging or cracking skin, altering its PH, or changing its normal flora- Recent studies suggest that frequent application of hand lotion may reduce this damage; medical personnel, therefore, are encouraged to use it. This rnay be difficult, however, as one should avoid applylng hand lotion immediately after washing, right before giving.direct patient care, or right before handling of sensitive instruments or equipment. Lotions might interfere with the residual action of the antimicrobial hand washing products.
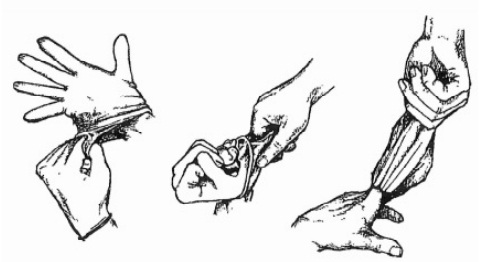
GLOVE REMOVAL
Hands will always be washed following glove removal. Gloves must be removed in a way that will avoid contamination of hands. Remove gloves when they become contaminated, damaged, between patients or before leaving the work area. With both hands gloved, peel one glove off from top to bottom and hold it in the gloved hand.
With the exposed hand, peel the second glove from the inside, tucking the first glove inside the second.
Dispose of the entire bundle promptly.
Wash your hands thoroughly.
PERSONAL PROTECTIVE EQUIPMENT (PPE)
The Employer must determine what personal protective equipment is needed for specific tasks performed in facility and suppiy the employee with all the necessary PPE (in appropriate sizes) for the tasks the employee performs. Employees are required to use PPE whenever there is a potential for direct skin contact with blood, hazardous chemicals, and potentially infectious materials.All personal protective equipment shall be removed immediately upon leaving the work area or as soon as possible and placed in an appropriate laundry hamper or disposed of. The employer is responsible for the laundering, cleaning and/or replacing of all PPE. It is the employers responsibility to enforce and monitor the use of PPE
POLICY: All employees will use the PPE recommended by the employer when performing procedures with potential contamination.
GLOVES
Gloves are available in all patient and testing areas of the facility. They are provided in sizes appropriate to the employees hand size. Hypoallergenic gloves will be provided or an alternative if latex sensitivity is a problem.
POLICY: Gloves will be worn when:
touching laboratory specimens
touching contaminated items
touching patient mucous membranes or non-intact skin
performing phiebotomy
when performing any surgical or invasive procedures
when performing laboratory tests
situations where hand contamination is possible
finger or heel sticks are performed
when handling contaminated laundry
when handling biohazardous waste or waste containers
when cleaning contaminated surfaces
when cleaning up spills
when the employee has breaks in the skin, including cuts, scratches, and chapped hands
GENERAL POLICIES:
Gloves must be changed between patients. Gloves must be removed before touching non-contaminated items, such as telephones.
Gloves (used) witl be disposed of according to local, state or OSHA regulations.
Gloves are not required by law to be used when administering intramuscular or subcutaneous
injections as long as bleeding the could result in hand contact with blood is not anticipated.
CLOTHING PROTECTION
Lab coats and clothing protection will be fluid repellent if there is a potential for splashing or spraying of blood or body fluids. Clothing protection will be fluid resistant (impervious) if there is the potential of soaking of blood or body fluids. Lab coats wom while performing laboratory tests are to be worn only in the laboratory area. The type and characteristics of the labcoatlapron/smock will depend upon the task and the degree of exposure anticipated. In areas where chemical splashes are great an impervious apron appropriate for the task shall be worn. Cloth labcoats worn to protect clothing from day to day soiling or to look professional, will only be used in situations where no contact with body fluids is expected. Lab coats, aprons or clothing cover must be worn when performing lab tests or procedures that splashing spraying or splattering is expected.
Soiled lab coats or clothing protection will not wom outside of the lab or procedure area.
Lab coats and clothing protection must be changed whenever visibly contaminated with blood or body fluids.
Disposable lab coats and clothing protection will be discarded in the biohazardous materials bag after use.
Contaminated reusable lab coats and clothing protection will be placed in the biohazardous laundry bag.
Lab coats and clothing cover will be fluid resistant and appropriate for the task involved.
Open back or open toe shoes may not be wom when performing procedures that may involve splashing or spraying of infectious materials.
MASKS, EYE PROTECTION, AND FACE SHIELDS
Masks and eye covering or a face shield will be worn during procedures that are likely to produce splashes, spray, splatter, droplets, or aerosols of blood or other potentially infectious materials. Contact lenses may not be worn without safety glasses when performing procedures with risk or when performing laboratory procedures with potential risk. Appropriate precautions must be applied and wearers alerted to potential hazards.Goggles or face shields are used when performing the following tasks in this facility:
______________________________________________________________________
______________________________________________________________________
______________________________________________________________________
______________________________________________________________________
Goggles and face shields will be cleaned after use and before storage. This facility uses the following method and product to clean goggles and face shields:
______________________________________________________________________
______________________________________________________________________
______________________________________________________________________
ACCIDENTAL NEEDLE STICKS & BODY FLUID EXPOSURE
POLICY: Health care workers will follow universal and standard precautions when handling blood and body fluids. If an exposure occurs, it will be reported immediately and acted upon promptly by the safety officer or employer.Exposure to blood and body fluids can be categorized as either percutaneous injuries (skin punctured by needle, scalpel, or other sharp object), mucous membrane splash (splash in the mouth, nose, or conjunctiva), or by open-wound contact (fresh open abrasions, incisions, human bites, or lacerations). Exposures by any body fluid to any open site will be taken seriously. Appropriate first aid will be administered as soon as possible. The incident will be reported to the safety officer or employer. The incident will be documented. The incident report will include detailed information on the accident. A form to document the exposure injury is found in the “Record Keeping” section of this manual. The employee will be sent to an appropriate facility for evaluation - as soon as possible after exposure.
Needle sticks account for 80% of all exposure injuries reported. The following policies will help prevent or reduce needle stick injuries:
Do not recap, remove, bend or break needles from disposable syringes. If recapping is unavoidable, use a scooping technique or mechanical device.
If syringes need to be taken to another area of the facility for disposal - place uncapped syringes in a secondary, covered transport container.
Discard used needles immediately after us by dropping needle down into sharps container - needle down
Do not overfill, shake or bang sharps containers.
If a needle starts to roll off a table or counter - DO NOT try to catch it. Let it fall to the floor and then pick it up.
Use safety needles and safety devices.
EXPOSURE FOLLOW-UP POLICY
Any direct exposure from needle stick or sharps: (1) make the injury bleed (optional) (2) immediately wash the area with antibacterial soap and flushed with large quantities of water (3) put antiseptic (betadine, iodine) on site.Any splash to the eyes or mucus membranes: (1) flush with large amounts of water at the eye wash station.
Report the injury to the safety supervisor - immediately or as soon as possible. Fill out an exposure incident form (found in record keeping section of this manual). This form contains information on route of exposure and circumstances under which the exposure incident occurred.
Arrange medical evaluation and treatment with a qualified health care provider as soon as possible or within 2 hours (CDC recommendation).
The source individual will be identified (if possible). The source will be asked to sign a consent. A consent form is found in the record keeping section of this manual. The sources blood will be tested, at no cost to individual, for presence of HIV, HBV and HCV. If the status of the sources HIV, HBV and HCV is known, it need not be repeated unless source is a high risk. If consent is not obtained, the employer shall establish that legally required consent cannot be obtained. If consent is not obtained the follow-up treats the source as positive for HIV, HBV and HCV. Results of the source individuals test will be made available to the exposed employee.
The employee will be counseled, informed of applicable laws and given a copy of the Bloodborne Pathogen Standard as part of the follow-up.
The employee will be provided information on the identity and infectious status of the source. ‘fhe employee will be vaccinated for HBV if not already vaccinated. If the employee is already vaccinated, the employee will be checked for current immunity status.
The employee will be asked to consent to having her/his blood collected and tested for HIV, HBV (or antibody status) and HCV. Collect blood as soon as possible (ie. within 2 hours) after the exposure. If consent is given for collection but consent is not given for testing, the blood will be preserved for at least 90 days. If the employee elects to have the blood tested during this period, it will be done as soon as feasible. The employee has the right to refuse blood collection and testing. If so, the employee will fill out the “Waiver of Exposure Evaluation” form found in the “Record Keeping” section of this manual.
If the results for HIV are sero-negative, the test will be repeated at 6 weeks, 12 weeks, 6 months and one year. The extent of the follow-up will be determined by the health care provider seeing the employee for the exposure. This information is confidential.
The administration of post exposure prophylactic treatment will be determined and recommended when medically indicated by the health care professional seeing the exposed employee. If recommended it will be started as soon as possible.
The employee will be given instructions on their responsibility for follow-up medical treatment/appointments and where they are to go to be evaluated.
All information relevant to the appropriate treatment, including vaccination status of the employee are to be provided by the employer to the evaluator.
A written opinion from the health care professional seeing the exposed employee will be provided to the employer and employee within 15 days. This option will not contain confidential information on the employee.
The employer will only be informed of the employee’s Hepatitis status, that the employee kept appointments for exposure follow-up, the employee was informed of the results of evaluation and that the employee has been told about any medical conditions resulting from the exposure.
ALL FINDINGS WILL REMAIN CONFIDENTIAL. An employee has a right to a private post-exposure follow-up. They cannot be forced to have this follow-up provided by the employer.
All record keeping will be maintained in a confidential manor by the employer.
A1l testing and treatment is at no cost to the employee.
The CDC “Hotline” phone # is 1-888-448-4911. Call this number to get current information on how to counsel and treat health care worker’s exposed to bloodborne disease.
BODY FLUID SPILLS
POLICY: All body fluid spills will be handled as potentially infectious material. The appropriate procedure for decontaminating spills of blood, body fluids, or infectious materials will include:Wear heavy duty gloves. Do not remove any glass or other share objects with your hands.
Rigid cardboard and a dust pan may be used to handle scoop up such objects.
Absorb the spilled material with disposable absorbent materials, such as paper towels or gauze pads.
Clean all visibly spilled material, using a detergent or disinfectant.
Flood the spill site, or wipe down the spill site with disposable towels soaked in disinfectant or l0o/o bleach solution (made fresh daily).
Wipe off the spill site with a clean dry paper towel or gauze pad. Alternately, the disinfectant may be permitted to air dry. follow manufacturer’s recommendations.
Place contaminated gloves and paper towels in a plastic biohazard bag and dispose according to medical waste policy. Wash hands appropriately.
Replace items used in spill kit so it will be ready to use next time needed.
Notes:
Report large spills to safety officer, laboratory director or employer before cleaning up.
Commercial spill kits may be purchased, if desired.
The spill kit(s) for facility is found:
______________________________________________________________________
______________________________________________________________________
______________________________________________________________________
______________________________________________________________________
SATETY FOR CARDIOPULMONARY RESUSCITATION (CPR)
Employees should be prepared for emergency situations when mouth-to-mouth resuscitation will possibly be necessary. It is wise for all people working in the medical field to take a Cardiopulmonary Resuscitation course. These cot uses are given at the YMCA, Red Cross and often at local hospitals and high schools. Mouth-pieces, resuscitation bags or other ventilation devices will be available to the staff. Employees will be instructed on their use. The resuscitation devise will be disinfected with 10% bleach or equivalent after use or discarded. Disinfectant such as 10% bleach will be left on for a minimum of 1 minute before being wiped off with a clean absorbent material.Although there has been no documentation of transmission of hepatitis B virus during mouth-to mouth resuscitation, theoretically there is risk of salivary transmission of HIV. As an additional precaution, gloves will be wom to prevent contact with blood or saliva in the patient’s mouth or on the face. CPR is not An OSHA regulalitions If facility has an AED - call POC to order a supplement for AED’s.
POLICY: Resuscitation devices are readily available and accessible to employees who can reasonably be expected to perform resuscitation.
Resuscitation devises are kept:__________________________________________
Copy of CPR training card will group was trained.:
be put in employee file.Enter employees certified in CPR or date group was trained.:
______________________________________________________ date _________
______________________________________________________ date _________
______________________________________________________ date _________
______________________________________________________ date _________
______________________________________________________ date _________
______________________________________________________ date _________
______________________________________________________ date _________
______________________________________________________ date _________
______________________________________________________ date _________
______________________________________________________ date _________
Use additional note book paper to document if more lines are needed.
FACILITY CLEANING POLICY
POLICY: The employer will assure that all work areas exposed to potentially infectious materials are disinfected and decontaminated as needed throughout the work day. Decontaminating procedures will always be performed prior to the general cleaning done by the housekeeping service.The method of decontamination will be based upon the location, type of surface to be cleaned, type of soil present, and tasks or procedures being performed in the area. The work site will be maintained in a clean and sanitary condition as outlined below:
1. All surfaces exposed to any body fluids in the examining/treatment room(s) and laboratory (if applicable) will be cleaned with an appropriate hospital grade disinfectant (EPA approved) at the end of each workday or as needed during the day. If l0% bleach is used it will be made daily or made and discarded after use as needed.
2. Examining room floors and laboratory floors (if applicable) will be cleaned a minimum of biweekly with a disinfectant cleaner.
3. Centrifuges (if present) will be wiped off with disinfectant weekly or as needed.
4. Phones will be frequently wiped off with disinfectant.
5. Any body fluid spills or chemical spills will be cleaned up immediately using the guidelines found in this manual. Employees will be instructed on the use and location of spill kits.
6. Medical procedure equipment and/or laboratory instruments that needs servicing will be decontaminated best as possible prior to having the service company make repairs.Service people will be instructed to wear gloves while servicing.
7. Protective coverings such as plastic wrap, aluminum foil, or imperviously-backed absorbent paper must be used to cover equipment and environmental surfaces. Coverings will be removed and replaced as soon as feasible after contamination.
8. Additional Cleaning Protocols specific to procedures performed in the facility such as cold disinfecting and heat sterilization are found elsewhere in this manual or have been written on the “Safety for Procedures Performed “ form found in this chapter.
FACILITY USES THE FOLLOWING PRODUCTS TO
DISINFECT WORK AREAS:
_______________________________ ______________________________________________________________ _______________________________
_______________________________ _______________________________
HOUSEKEEPING
EMPLOYER/EMPLOYEE POLICY: It is the employers responsibility to ensure that the facility is maintained and cleaned in a sanitary manner. If any surface is contaminated during the day, it will be cleaned and decontaminated immediately by the staff using a “hospital grade” (EPA approved) disinfectant. Spill kits and protective covering will be available to the staff. The staffwill be instructed on how to decontaminate work areas and clean up spills (chemical and biohazard). These cleaning procedures will always be done BEFORE the hpusekeeping service comes tg clean.HOUSEKEEPING SERVICE POLICY: Housekeeping workers will be made aware that certain areas ofthe facility might be contaminated with potentially infectious materials. Housekeeping workers will wear all purpose utility gloves and protective clothing when cleaning these areas. Housekeeping workers will be instructed on where infectious waste (biohazard bags and sharps containers) is found and where (if applicable) it is stored prior to pick up. The housekeeping service will use an appropriate level I disinfectant to routinely clean work surfaces and floors. An EPA approved level 2 disinfectant will be used to clean operating rooms. Walls, blinds and curtains only need to cleaned if visible soiled.
CARPET CLEANING POLICY: If the facility has carpets, the carpets will be cleaned on a regular bases with a disinfectant or germicidal type rug cleaner. Carpets are not recommended for examining rooms and laboratory areas.
POLICY FOR HOUSEKEEPING SERVICE:
This facility uses the following Cleaning Service:________________________________The Service cleans the facility (frequency):____________________________________
The Service has been instructed on disinfectants to be used: yes_ no_
The Service workers are aware of safety precautions: yes_ no_
The Service workers understand bio-hazard labels: yes_ no_
The Service uses PPE when cleaning facility: yes_ no_
The Service is aware of areas they should not clean or touch yes_ no_
COMMENTS:__________________________________________________________
_____________________________________________________________________
New Housekeeping Service: ____________________________________________
Date:____________
New Housekeeping Service: ____________________________________________
Date:____________
LAUNDRY
The Bloodborne Standard specifically addresses laundry practices in the faciiity.POLICY: The employer will ensure the following:
1. Contaminated laundry will be bagged at the location where is was used.
2. Contaminated laundry will be placed and transported in bags that are labeled or color coded and will prevent liquid seepage if such a potential exists. Employees will be informed about the labeling system.
3. Laundry that is contaminated with blood or other potentially infectious materials will be treated as infectious, handled as liule as possible and gloves worn while handling.
4. If a laundry service is used, the laundry workers will wear protective gloves to prevent exposure to potentially infectious materials while sorting and handling.
5. If employees have contact with contaminated laundry they will wear protective gloves as well as any other appropriate personal protective equipment while handling it.
6. Handle soiled linen as little as possible and with a minimum of agitation to prevent gross microbial contamination of the air and of the persons handling the linen.
7. Linens are to be washedwith detergent inhotwaterofat least 71"C (160"F) for 15 - 25 minutes.
8. If a lower temperature is used a chemical such as bleach must be added.
9. If lab coats, aprons or smocks are used by employees as personal protective equipment, the employer shall provide forthe cleaning and laundering. Personal protective equipment shall be removed prior to leaving the work area and placed in appropriately designated area or container, prior to washing. Lab coats used only to look profession (not for protection) may be taken home by the employee to launder.
Home Laundering:
Home laundering of contaminated PPE worn by employees is not permitted since the employer cannot guarantee that proper handling or laundering procedures are being followed. When not done correctly it may lead to the migration of the contaminants to the home laundry.If the employee wishes to choose, wear, and maintain his/her own uniform or laboratory coat, then he/she would need to don additional employer supplied PPE and employer controlled PPE when performing tasks where it is reasonable to anticipate exposure to blood or body fluids.
COMMENTS:________________________________________________________
___________________________________________________________________
LAUNDRY POLICY FOR FACILITY
___________ Check here if facility uses all disposable linens for patients___________ Check here if only laundry sent out is physician or staff lab coats
LAUNDRY SERVICE POLICY: All soiled laundry or linen will be handled as if potentially contaminated with body fluids.
All clean linen will be stored in an area where no soiled linen will come in contact with it.
Clean linens are stored:__________________________________________
All soiled laundry or linen will be bagged at the site where it is used. It will be placed in a laundry bag that can be identified as follows:__________________________________
Linen bags with soiled laundry are stored:______________________________________
This facility uses the following laundry service:__________________________________
Laundry is picked up:___________________________________________________
Laundry service has been instructed on the handling of facility linen: yes____no_____
date of instruction:__________________________
If the facility does the laundry on site, the following laundry procedure is followed:
______________________________________________________________________________
______________________________________________________________________________
______________________________________________________________________________
The person designated to do the laundry is:____________________________________________
_____________________________________________
PPE will be used by any person who handles or launders soiled laundry
If laundry service changes - write in new name and date below:
Date : _________ Name: ______________________
_________ ______________________
_________ ______________________
STERILIZATION & DISINFECTION
Many agents are available to kill microorganisms. You must choose the agent appropriate for the type of item or surface it will be used on. It is the responsibility of the staff to keep all work area’s clean and disinfected throughout the workday. Disinfection and sterilization procedures should be used for all reusable instruments, devices and other items that are contaminated with blood and/or other potentially infectious materials (OPIM). Always follow the manufacturer’s instructions.
Antiseptics are chemical germicides that are formulated to used on skin or tissue. Antiseptics WILL NOT BE USED TO DECONTAMINATE INANIMATE OBJECTS OR SURFACES.
Disinfectants are agents that destroy or inactivate specific viruses, bacteria, or fungi on inanimate surfaces. Disinfectants come in three levels; low, intermediate and high. Select the appropriate level for the item(s) you are disinfecting. Check with the manufacturer if you are unsure.
Level 1 - Low: Appropriate for non-critial care items that have not been contaminated with blood or OPIM. Good for floors, counter tops, crutches, etc.
Level2 - Intermediate: To be used to disinfect non-critical care items that have been contaminated with blood or OPIM. Good for potentially contaminated surfaces such as a phlebotomy chair or counter where blood tests are performed.
Level 3 - High: To be used for all semi-critical care items that could be damaged by heat sterilization.
Time needed to soak these item can be found in the manufacturer’s instructions for use. Semi-critical items would be instruments that touch mucous membranes but do not penetrate tissue. This level would be cold sterilization solutions.
Sterilization: A process that destroy all viruses, bacteria, fungi and their spores. This would include instruments and/or devices that are introduced directly into the bloodstream or penetrate tissue.
Sterilization should be used on all critical care items that can withstand the sterilization process.
Sterilizing methods include use of steam under pressure (steam autoclave), gas, dry heat or chemicals under pressul’e (chemical autoclave).
Qualify Control: Sterilization and level 3 disinfecting methods must be monitored for effectiveness.
Controls are corrmercially available. Controls are a valuable way to determining whether or not the conditions of the sterilization have been achieved. Both chemical indicators (color indicator tape) and biological indicators (spore tests) will be used as recofllmended by the manufacturer.
TYPES OF DISINFECTANTS
Intermediate-level (level 2): Disinfectants the EPAterrns as “hospital grade” and have been tested and shownto destroy TB, HBV or HIV. Intermediate level disinfectants will be used to decontaminate non critical items that have contact with blood and body fluids. A 1:10 bleach solution is recommended by OSHA. If l0% bleach is used it must be made fresh daily.Product(s) used:_____________________________________________
Product(s) used:_____________________________________________
(1) Cold sterilization High level disinfectants with glutaraldehyde that are used on reus able semicritical items that come in contact with potentially infectious body fluids. Items will be immersed an soaked according to the manufacturer’s instructions. Control test strips will be used daily after 2 weeks to check effectiveness of disinfectants used for 28 days before disposal.
Product(s) used:_____________________________________________
Product(s) used:_____________________________________________
(2) Heat Sterilizatron:Steam autoclave or dry heat autoclave.
Type used:_____________________________________________
(3) Other:_____________________________________________
All reusable critical care items that can be sterilized - WILL be sterilized after disinfecting.
Reminder: Make sure you have an SDS sheet for all level 2 and 3 disinfectants!
SECTION 5 REVISED GLOBAL HAZARD COMMUNICATION STANDARD
(“RIGHT TO KNOW”)
AND
‘’ OCCUPATIONAL EXPOSURE STANDARD’’
(CHEMICALS IN LABORATORIES)
THE REVISED GLOBAL HAZARD COMMUNICATION STANDARD
The revised Global Hazard Standard was released in the Code of Federal Regulations for Labor on March 20, 2012. A copy of law is found in chapter 2 of this manual. The standard addresses potentially hazardous chemicals in the workplace. The revised standard will be updated every 2 years. The 2012 revised law is a revision of the 1989 Hazard Communication Standard. The reason for the update is to align the OSHA standard with a global chemical standard called the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). This GHS was developed by the United Nations. Chemicals will be classified according to their health and physical hazards (there will be 10 health hazards and 16 physical hazards). What is the Globally Harmonized System of Classification and Labeling of Chemicals (GHS)? It is an internationally accepted system of classification and labeling of chemicals. United States adoption of this system will help manufacturer’s classify, their products for local
and international distribution.POLICY: The employer will insure that employees have a safe and healthful work environment when working with chemicals.
The Hazard Communication Standard covers employee physical and health hazards related to chemicals found in the medical facility. The purpose is to ensure that employees be informed about the particular hazardous substances in their work place, the health risks associated with them, and how to take protective action. It requires the following:
1. A written Hazard Communication Program that evaluates potential chemical hazards and communicates information concerning these hazards. Plan will be reviewed and upgraded annually.
2. Developing policies on protective measures to be used.
3. Having procedures in place to follow if an exposure to a chemical occur
4. Safety Data Sheets (SDS) for all hazardous chemicals found in the work place. SDS’s must be inventoried, indexed and reviewed annually.
5. A hazard labeling system that provides an immediate warning for employees working with hazardous chemicals. (see “Label” section of manual)
6. Documented employee training on chemical safety.
7. Proper storage of chemicals.
8. Posting of OSHA required documents, posters and reports.
Inspection For This Standard: The best defense to avoid OSHA violations and fines is to know the law and be prepared by providing employees with a safe and healthful work environment as specified by OSHA. An OSHA inspection of the work place can occur when an employee, patient or appropriate representative files a complaint with OSHA. If the work place has more than ten employees it can be subject to an administrative inspection by OSHA without a complaint. The facility does have the right to refuse an inspection if the safety officer or employer is not on-site (this at best only buys the facility a brief amount of time before the inspector returns). The inspection may not be refused if the OSHA representative has a legal warrant to conduct an inspection.
 Hours:
Hours: