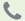BIOHAZARD LABELS

The Bloodborne Standard makes it mandatory that containers which hold or store biohazardous materials must be properly labeled. Biohazardous materials include blood and any potentially infectious body fluid. Receptacles that hold contaminated needles,
gloves, masks, gauze, wipes, garments, etc., must be display a bio-hazard label. Labels shall be used to identify the presence of an actual or potential biological hazard.
CONSIDERATIONS:
Labels shall be fluorescent orange or orange-red, with lettering or symbols in a contrasting color.
Labels will be affixed on to or as close as feasible to the container by adhesive, string, wire or other method.
All infectious waste containers will be labeled with a biohazard label. Red bags or red containers may be substituted for labels if staff has be trained on this system.
If blood or control serum is stored in refrigerator or freezer, the refrigerator/freezer shall be marked with a biohazard label.
If blood is stored in refrigerator for transport it will be put in containment bags marked biohazard.
If blood is placed in a pick-up container supplied by the reference laboratory - the container will be marked with a biohazard label.
If body fluids are transported to another location by facility or courier, the specimens will be double bagged, the outer bag marked biohazard, and the transport container
will be marked with a biohazard label. Cooling packs will be used to maximize specimen integrity while being transported.
CHEMICAL- WARNING LABEL DETERMINATION
The Global Hazard Communication Standard mandates specific chemical labeling requirements. Labels must be on all hazardous chemicals that are shipped to and used in the work place. Labels must not be removed. Safety Data Sheets (SDS) for all chemicals will be available to employees.Manufacturer Requirements: Chemical manufacturers are required to evaluate chemicals, determine status as hazards, provide Safety Data Sheets* and properly label all shipped chemicals. Manufacturer labels must never be removed. The best way to determine the hazard of the chemical is to read the SDS or obtain an OSHA designated list or Hazardous Substance list. The term danger is used for extreme hazards and warning is used for less severe hazards. Label must contain a precautionary statement. Pictograms (picture warnings) may be used to alert the user on the label. For most “mixed” chemicals it is necessary to contact the manufacturer or supplier for the SDS.
Safety Data Sheets (SDS): Safety Data Sheets (SDS) are manufacturer information forms with extensive information on the contents of the product and safety precautions to take when using that product. Additional information on SDS’s can be found in the “Hazard Communication” chapter of this manual. The employer will compile an SDS file or notebook. This notebook or file will contain a chemical inventory of all SDS’s. The notebook or file will be reviewed and updated annually or as needed. The SDS notebook or file will be located in a place where it is accessible to all employees.
Information on SDS’s and chemical labeling will be provided during the Safety Training Program.
Facility Chemicals: Search through your facility and write down all chemicals you have in facility. Most pharmaceutical, cosmetic and “common” household products are exempt from this standard unless they are used in a way or in a quantity that would not be considered “every day” normal use. Ingredients can then be compared to a list of regulated substances published by OSHA or the SDS will provide necessary information.
* Under the revised standard manufacturers had until June 1.2015 to change the material safety data sheet (MSDS) format to the new SDS format.
Employers Responsibility: Any hazardous chemical used in the work place that is not in its original container or missing a label must be labeled with identity of the chemical and hazards. There are different types of chemical labels available; NFPA, NFPA with target organ, and commercially made labels. A facility may also copy a manufacturer label and attach it to the unmarked container. The labels must include the chemical and/or common name and warnings about physical, fire and health hazards.
Exceptions to labeling requirements: If a chemical is removed from the original container and put into a secondary con tainer for immediate use - the secondary container does not need to be labeled.
If a chemical is removed from the original container and put into a secondary container and used during the work shift and discarded at the end of the day - it does not need to be labeled.
Exceptions to SDS’s:
Most pharmaceutical do not require an SDS. An exception would be pharmaceuticals with a potential danger to the employee working with them. Examples would be chemotherapy drugs, acids used for procedures such as wart removal and live vaccines.
Most household products do not require SDS’s. An exception would be household product not used as intended or a product used more frequently than intended. Examples would be bleach being used as a disinfectant or a cleaning product being used continually during the work day.
If in doubt, ask the manufacturer. It is better to do more than you need to do than to do less!
WARNING PICTURES/SIGNS/LABELS
Warning signs and symbols for potential dangers are located in many areas of the clinical facility. Employees may see these signs on doors, refrigerators, storage cabinets, reagent bottles, and Safety Data Sheets (SDS). Employees will be able to recognize each one for its specific danger and the necessary precautions. Under the revised Hazad, Communication Standard of 2012 these “pictograms” symbolize the following
hazards:
PICTOGRAMS AND HAZARD CLASSES UNDER THE REVISED STANDARD

NFPA CHEMICAL HAZARD SIGN

This special label warns emergency, rescue, and fire personnel of potential hazards within a specified area and allows them to take necessary precautions. The numerals used (0-4) are from the National Fire Protection Association (FPA) hazard rating system. This labeling system can be used to label chemicals that have been removed from manufacturer’s containers in the facility.
The numbers, which indicate levels of risk for each product, are assigned by the manufacturer or can be assigned by the facility using this rating system after reviewing the SDS. The NFPA hazard sign is often seen on commercial product labels, however, it is not required if the manufacturer has provided verbal or picture warnings. If this facility uses this labeling system, a poster explaining the colors and codes will be displayed for employees. A poster to display is found in the front cover of the manual - post if NFPA labels are used in facility.
HOW TO READ A NFPA LABEL
The blue area warns of a health hazard according to the following number code:
4. Danger: Extreme. Deadly. Highly toxic. May be fatal on short exposure. Specialized protective equipment required.
3. Warning: Serious. Corrosive or toxic. Avoid inhalation and/or skin contact or Inhalation.
2. Warning: Dangerous. Moderately toxic. May be harmful if inhaled or absorbed.
1. Caution: Slightly toxic. May cause irritation.
0. No unusual hazard.
The red area warns of a fire hazard according to the following numbercodes:
4. Danger: Severe. Extreme. Very flammable. Flammable gas or extremely flammable liquid. Flash point <73° F.
3. Warning: Serious. Extreme Caution. Flammable liquid. Flash point 73-100°F.
2. Caution: Moderate. Caution. Combustible liquid. Flash point of 100° F to 200°F.
1. Slight: Combustible if heated.
0. Not combustible. Will not burn.
The yellow area warns of a reactivity or instability hazard according to the following
number code:
4. Danger: Extreme. Severe. Explosive material at room temperature.
3 . Danger: Serious. Explosive. May be explosive is spark occurs, if heated under confinement, or mixed with water.
2. Warning: Unstable, or may react if mixed with water.
1. Caution: Slight. Normally stable. May react if heated, or mixed with water.
0. Stable: Not reactive when mixed with water.
Example 1:
A .Goggles/safety glasses
B. Goggles, Gloves
C. Goggles, Gloves, Apron
D. Face Shield, Gloves, Apron
E. Goggles, Gloves, Mask
F. Goggles, Gloves, Apron, Mask
G. Gloves
Example 2:
SG Safety Glasses
R.P Respiratory Protection
GL Gloves
FS Face Shield
VA Vinyl Apron
LC Lab coat
NOTE: Additional coding options are available, but are usually not relevant to a physician office. These options can be found on the Hazard Material Identification Guide Sticker found in the front sleeve of this manual.
If NFPA labels are used. a poster with instructions on how to read a NFPA label will be posted in facility. Safety training will include how to read a NFPA label.
TARGET ORGAN LABELS
Target organ labels should be used if an exposure to a chemical product can result in damage to an organ. Target organ labels are available from safety supply companies and are easy to use. They contain a list of organs than can be damaged and you simply check the ones that apply. The information needed is found on the SDS.

SUMMARY
The EMPLOYER is responsible to ensure that any container containing a hazardous chemical is labeled with the chemical identity and hazard warnings.
The name on the label must match the name of the inventory sheets and SDS.
Labels must be in English, legible, and prominently displayed.
Staff training is required to ensure comprehension of labels and SDS’s for hazardous chemicals.
Outdated chemicals will be disposed of properly.
Chemical inventory will be reviewed and updated annually.
Any containers which hold or store blood will be marked with a Biohazard Label.
Any containers that store potentially contaminated/infectious materials will be marked with a Biohazard Label.
LABELS CAN BE ORDERED FROM:
(1) POINT OF CARE CONSULTS 1-856-580-0801 - 10 labels for $1 plus shipping charge
(2) “Grainger” l-800-GRAINGER - call to get catalog
 Hours:
Hours: